 sp 3 hybridization gives a tetrahedral molecular geometry.sp 2 hybridization has a triangular planner geometry.sp hybridization give a linear molecule.Thus an atom that has s and p orbitals in its valence shell can form 3 types of hybrid orbitals All four bonds of this molecule are sigma bonds and constitute sam lengths due to hybridization.Ĭarbon has one (s) and three (p) orbitals which are hybridized to form four new degenerate orbitals. Hybrid means a mixture of two which in molecular geometry states that two or more atomic orbitals of a central atom are mixed to form new and degenerate (same energy) hybridized orbitals.įor example, in a methane molecule, carbon is bonded with 4 hydrogen atoms which means that carbon has four atomic orbitals involved in bonding. In a localized bond approach after drawing a lewis diagram of a molecule, hybridization or VSEPR model is applied to understand its best representation. The combined or hybrid structure deduced from the possible lewis structures is given below: These possible structures are known as their canonical forms and are best described as hybrids of all the resonance forms. Some molecular compounds have more than one lewis diagram. Lewis structure of the resonating structures It may be noted that the best lewis diagram is the one with the least formal charge. Calculate the formal charge on each atomįormal charge = Valence electrons – (Bonded electrons + Non-bonded electrons)įormal charge on nitrogen = 5 – 3 – 2 = 0įormal charge on fluorine-(1) =7- 6 -1 = 0įormal charge on flourine-(2) =7- 6 -1 = 0įormal charge on flourine-(3) = 7- 6 -1 = 0.Place the remaining electrons on the central atomĪfter the connection and distribution of electrons, 2 electrons are left behind (from 26) which are placed on the central atom.Now complete the octet of all peripheral atoms. Complete the outermost electrons of peripheral atoms.The central atom is connected with other atoms through lines called bonds to give a skeleton to the molecule. Connect the central atom with surrounding atoms to draw the structure.Hence, nitrogen in NF 3 is the central atom here. Oxygen and hydrogen are generally peripheral atoms. The central atom should be the least electronegative one. It is a crucial step to draw the best lewis structure. 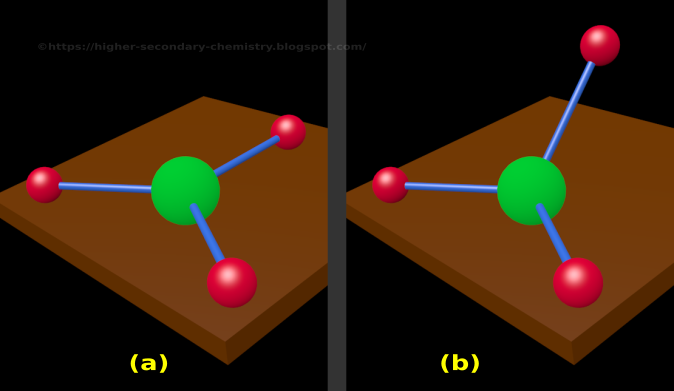
The valence electrons of nitrogen and the fluorine atoms are added and the number comes out is 26. Count the total number of valence electrons in a molecule.The lewis diagram of a molecule (NH 3) can be drawn by following the step below Example: Lewis diagram of (NF 3) molecule He was the first-ever scientist who presented the electronic structure of a molecule or a polyatomic ion.Įlectron and molecular geometry provide a complete framework of molecular geometries. According to him, two electrons shared by each atom to form a bond form a bonding pair, while the electron pair on each atom not involved in bonding is called a lone pair. He first recognized that bonding between two atoms is due to sharing of electrons. 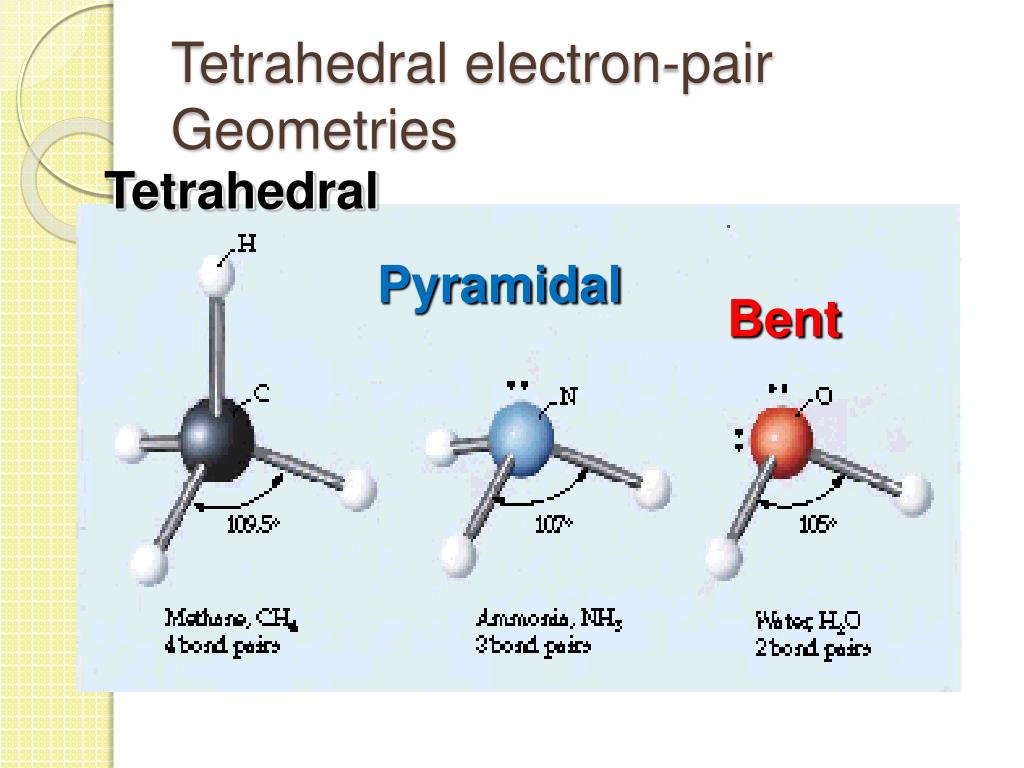
How to determine the molecular geometry.Lewis structure of the resonating structures.Example: Lewis diagram of (NF3) molecule.To determine the molecular shapes or geometry, three theories are to be understood and are to be somehow simultaneously applied. Covalent bonds are explained with a combination of theories of bonding. The localized bond approach is a simple and sophisticated bond that is easy to draw on paper. The bonding sense is really important to figure out the reactivity, polarity, and physical properties of molecules. They explain the electronic structure of molecules. 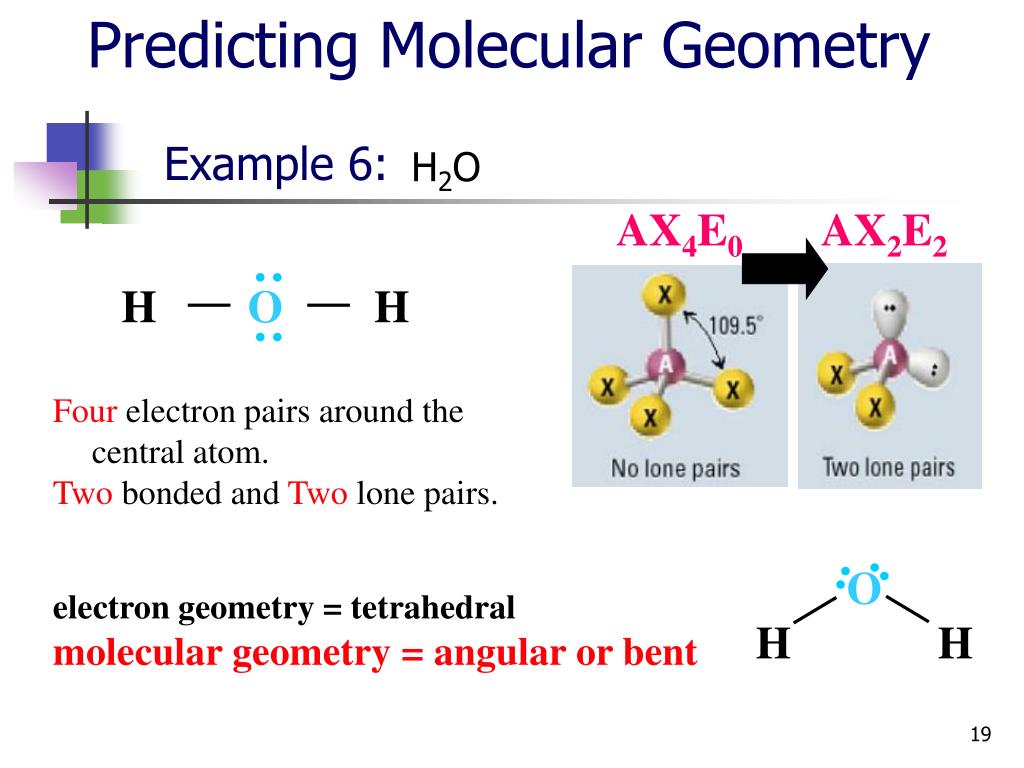
These theories about bonding though give incomplete knowledge but are still very useful. These techniques give data with great accuracy.īasic knowledge about bonding in molecules helps to understand their geometries such as Lewis theory, VSEPR theory, and hybridization theory. For example, x-ray diffraction and spectroscopy. There are some advanced techniques that help to understand the molecular geometry of molecules. Molecular geometry can be defined as the arrangement of atoms of molecules in a 3-dimensional space. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
